TSCA
Toxic Substances Control Act
TSCA is the primary U.S. federal law governing chemical substances. Administered by the EPA, TSCA requires that chemical substances manufactured, imported, processed, or distributed in the United States be evaluated for risks to human health and the environment. Recent amendments under the Lautenberg Chemical Safety Act (2016) have significantly expanded EPA's authority to evaluate and restrict chemicals.
What you receive
TSCA Compliance Certificates & Inventory Verification
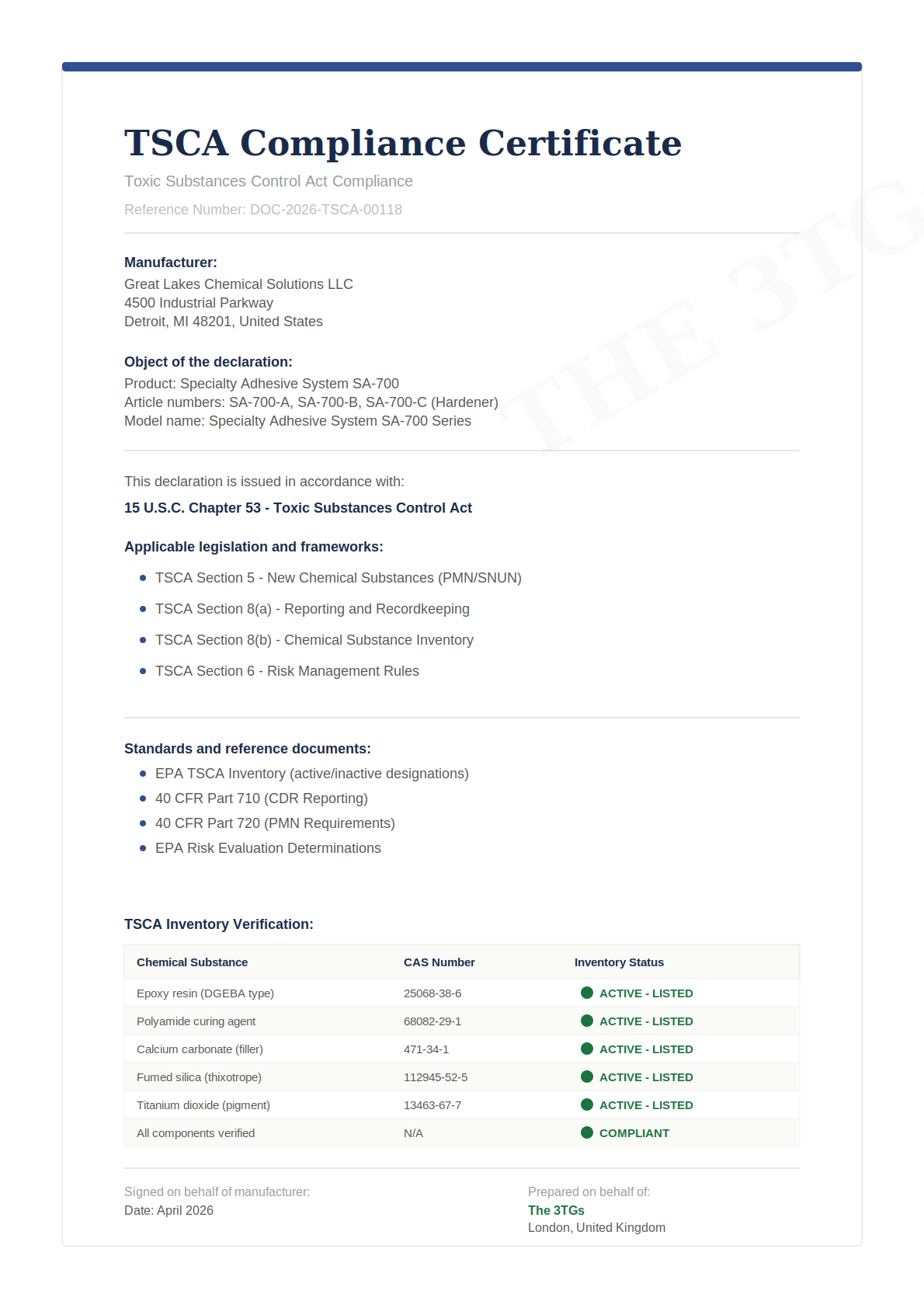
We deliver TSCA compliance documentation including Inventory verification for every chemical substance in your products, CDR reporting support, and ongoing tracking of EPA risk evaluations that may affect your product portfolio.
- TSCA Inventory status verification with CAS number documentation
- CDR reporting preparation and filing support
- Section 6 risk evaluation tracking for substances in your products
- Compliance records structured for EPA audit readiness
Manufacturers and importers of chemical substances and mixtures in the United States, as well as companies that process or distribute these substances. If your products contain chemical substances that are manufactured or imported into the U.S., TSCA obligations may apply to you, particularly around inventory compliance, reporting, and risk management rules.
- TSCA Inventory: all chemical substances manufactured or imported commercially must be listed on the TSCA Inventory
- New chemical review: substances not on the Inventory require pre-manufacture notification (PMN) before commercial production
- Chemical Data Reporting (CDR): manufacturers and importers must report production volumes and use data every four years
- Risk evaluations: EPA is conducting risk evaluations on high-priority substances under Section 6
- Section 6 risk management: EPA can impose restrictions, bans, or conditions of use based on risk evaluation findings
- TSCA Section 8(a)(7): one-time PFAS reporting requirement for historical manufacture and import
Key thresholds and timelines
- CDR reporting threshold: 25,000 lbs per site per year for most chemicals
- CDR reporting cycle: every four years (most recent: 2024 for reporting year 2023)
- EPA has identified 20+ high-priority substances for risk evaluation under Section 6
- Significant New Use Rules (SNURs) can trigger notification requirements for changes in how a substance is used
- Non-compliance penalties: up to $25,000+ per day per violation
- Not verifying whether chemical substances in imported products are on the TSCA Inventory
- Missing CDR reporting obligations for substances that cross the 25,000 lb threshold
- Not tracking EPA risk evaluations that may result in restrictions on substances in your products
- Ignoring Significant New Use Rules (SNURs) that may apply to new applications of existing substances
- Assuming TSCA only applies to chemical manufacturers and not to importers or downstream users
- Not maintaining records sufficient to demonstrate compliance in an EPA audit
- TSCA Inventory status verification for chemical substances in your products
- CDR reporting support and filing preparation
- Section 6 risk evaluation tracking and impact assessment for your product portfolio
- SNUR applicability review for new or changed substance uses
- Supplier data collection aligned to EPA reporting requirements
- Compliance documentation and audit readiness
- Ongoing regulatory monitoring for new EPA actions under TSCA
Need TSCA compliance support? We verify your inventory status, manage your reporting obligations, and track EPA actions that affect your products.
Contact Us →info@3tgs.org · +44 20 3996 3623

